What Is Delayed Sleep Phase Syndrome?
Delayed Sleep Phase Syndrome — known clinically as Delayed Sleep-Wake Phase Disorder (DSWPD) — is a circadian rhythm sleep-wake disorder in which a person's internal body clock runs on a schedule that is significantly later than conventional social norms. Someone with DSPS cannot fall asleep until very late at night (often 2–6 AM) and, if left alone, would not wake up until late morning or afternoon.
DSPS is not insomnia in the traditional sense. The person can sleep well once they fall asleep — the problem is the timing of sleep, not the ability to sleep. When forced to wake for school or work at a conventional hour, they end up chronically sleep-deprived, exhausted in the morning, and most alert in the evening when everyone else is winding down.
DSPS is the most common intrinsic circadian rhythm disorder. It affects an estimated 7–16% of adolescents and young adults, and roughly 0.2–1.7% of the general adult population, though milder forms are much more widespread.
New to circadian rhythms? If you want a foundational overview of the body clock before diving into DSPS, start with our general article on Circadian Rhythm Explained: Your Body's Internal Clock.
It Is Not Laziness or Poor Discipline
One of the most damaging misconceptions about DSPS is that it reflects a lack of willpower, poor sleep habits, or teenage rebellion. Decades of circadian biology research have established that DSPS is a genuine biological condition involving the master clock in the brain, the genes that regulate it, and the way the clock responds to light. People with DSPS are not choosing to stay up late — their internal clock is physically set to a later time than the external world.
The Science: How Your Body Clock Works
To understand DSPS, it helps to understand the hardware your brain uses to keep time.
The Suprachiasmatic Nucleus (SCN)
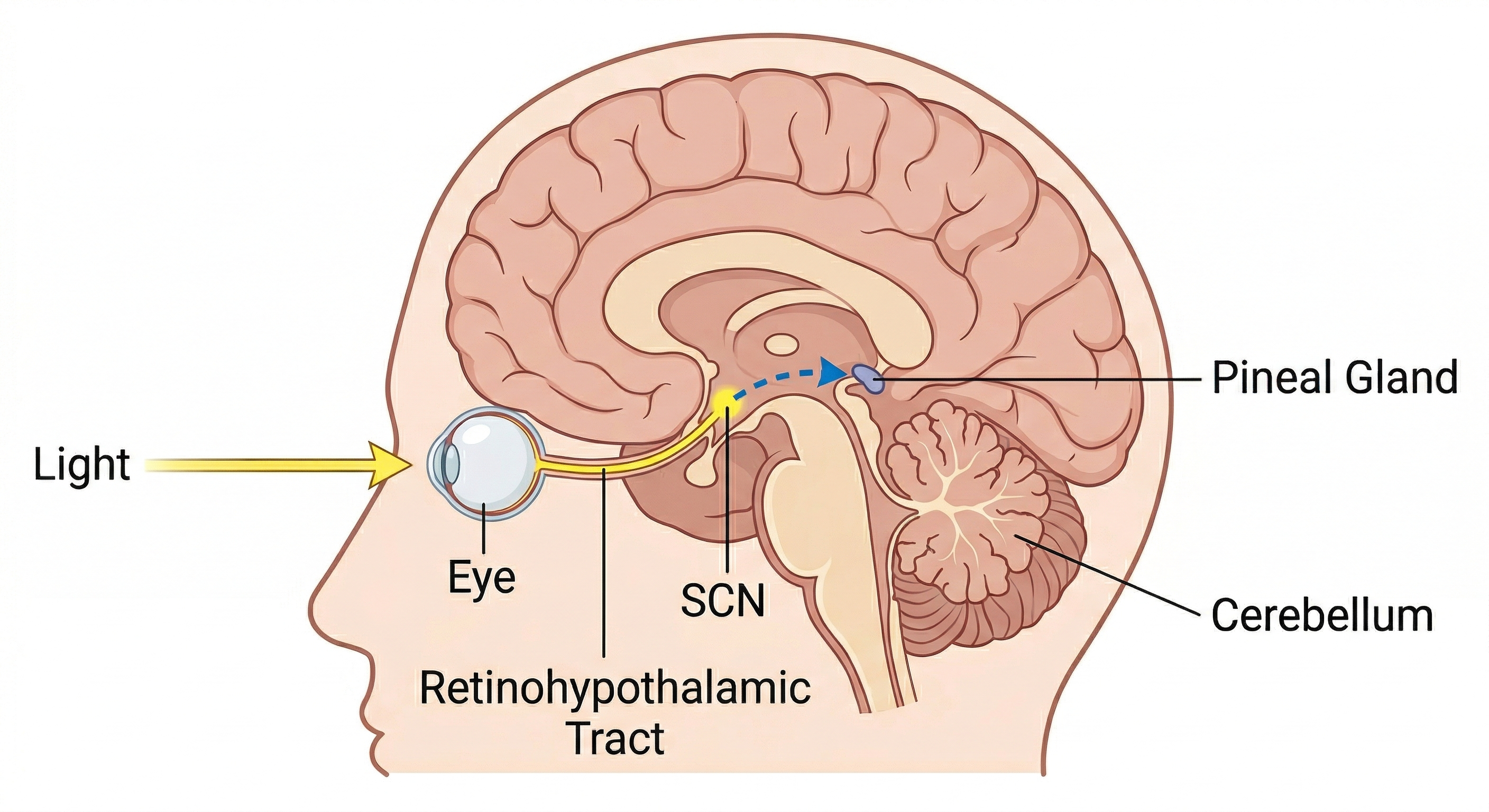
Deep inside the hypothalamus, just above the point where the optic nerves cross, sits a pair of tiny nuclei about the size of a grain of rice: the suprachiasmatic nucleus (SCN). Roughly 20,000 neurons in the SCN function as the body's master circadian pacemaker. They generate a self-sustaining ~24-hour rhythm through feedback loops of "clock genes" (such as BMAL1, CLOCK, PER, and CRY), and they broadcast timing signals to virtually every organ system in the body.
Left entirely in the dark, the human SCN runs on an intrinsic period slightly longer than 24 hours — typically about 24.2 hours. To stay synchronized to the real 24-hour day, it must be reset daily by external time cues. The strongest of these cues, by far, is light.
The Light Pathway to the SCN
Light reaches the SCN through a dedicated, non-image-forming pathway that is completely separate from the visual pathway you use to see. Specialized retinal cells called intrinsically photosensitive retinal ganglion cells (ipRGCs) contain a pigment called melanopsin, which is most sensitive to short-wavelength blue light around 480 nm. These cells project directly through the retinohypothalamic tract to the SCN.
When light hits the retina, ipRGCs fire and signal the SCN that it is day. The SCN then gates activity reaching the pineal gland through a multi-step neural pathway. In darkness, the pineal releases melatonin; in light, that release is suppressed. This is why melatonin is best described not as a sleep hormone but as a darkness hormone.
The Phase Response Curve
The same amount of light can either advance your clock (make you sleepy earlier the next night) or delay it (push you later), depending on when in your circadian cycle you receive it. The phase response curve (PRC) to light summarizes this:
- Light in the early biological morning — just after your core body temperature minimum (CBTmin) — advances your clock earlier.
- Light in the biological evening and early night — before CBTmin — delays your clock later.
- Light around the middle of the biological day has little phase-shifting effect.
Melatonin has the opposite PRC: low-dose melatonin taken in the biological evening advances the clock; taken in the biological morning it delays it.
These two curves are the scientific foundation of DSPS treatment.
What Delayed Sleep Phase Looks Like
A person with DSPS typically shows the following pattern:
- Persistent difficulty falling asleep at a conventional bedtime (e.g., lying awake until 3 or 4 AM).
- Great difficulty waking at the required time, with morning grogginess, irritability, and confusion.
- Excessive daytime sleepiness in the morning and early afternoon.
- Peak alertness and productivity in the evening and late night.
- Normal sleep architecture and duration when allowed to sleep on their own schedule — for example, on vacation or over a long weekend, sleep becomes effortless and refreshing as soon as bedtime shifts later.
- Symptoms persist for at least three months.
Diagnostic Criteria
According to the International Classification of Sleep Disorders (ICSD-3-TR), a diagnosis of DSWPD requires all of the following:
- A significant delay in the timing of the major sleep episode relative to the desired or required sleep and wake times, demonstrated by a chronic or recurrent complaint of inability to fall asleep and difficulty awakening at a desired time.
- The symptoms are present for at least three months.
- When patients are allowed to choose their own schedule, sleep quality and duration improve and a consistent but delayed schedule emerges.
- A sleep log and, whenever possible, actigraphy for at least 7 days (preferably 14) documents the delay in timing of the habitual sleep period.
- The disturbance is not better explained by another sleep disorder, medical or neurological disorder, medication, or substance use.
Polysomnography is not required to diagnose DSPS, but may be ordered to rule out comorbid conditions such as obstructive sleep apnea. A sleep specialist may also measure dim light melatonin onset (DLMO) from saliva samples to objectively establish the timing of the circadian phase.
Causes and Contributing Factors
DSPS is best understood as a mismatch between the internal clock and the social clock. Several biological and behavioral factors contribute:
Biological Factors
- Intrinsic circadian period longer than average. Some people's clocks naturally run closer to 24.5 or even 25 hours, which creates a constant drift toward a later schedule.
- Altered phase response to light. People with DSPS may be more sensitive to evening light (more delay) and less sensitive to morning light (less advance).
- Clock gene variants. Polymorphisms in PER3, CRY1, and related genes have been linked to extreme evening chronotypes and DSPS in family and genetic studies.
- Adolescent development. Puberty is associated with a natural, hormone-driven delay in circadian phase of about 1–3 hours. This is why teenagers genuinely need to sleep later — and it is why DSPS often first emerges in the teen years.
Behavioral and Environmental Factors
- Evening bright light (overhead LEDs, screens, gaming, late-night studying) that delays the clock further.
- Insufficient morning light — rolling out of bed into a dim, curtained house never delivers the morning light signal needed to reset the clock.
- Irregular weekend schedules that repeatedly push the clock later.
- Napping late in the day, which reduces sleep pressure at night.
- Caffeine late in the day, which blocks adenosine and sabotages sleep onset.
- Anxiety about falling asleep, which creates conditioned hyperarousal layered on top of the biological delay.
The behavioral factors do not cause DSPS in a person with a normal clock, but in someone with an underlying circadian predisposition, they can turn a mild tendency into a disabling disorder.
Consequences of Untreated DSPS
Because people with DSPS are forced to wake up on a schedule their biology does not support, the disorder commonly leads to:
- Chronic sleep deprivation on school or work days
- Academic and occupational underperformance
- Depression, anxiety, and mood instability (DSPS shows a strong bidirectional relationship with depression)
- Increased risk of substance use (caffeine, stimulants, alcohol as a sleep aid)
- Cardiometabolic consequences of chronic circadian misalignment
- Social and family conflict over perceived "laziness"
The good news is that DSPS is treatable — often dramatically so — when the biology of the body clock is addressed directly.
Treatment: Resetting the Clock
The 2015 AASM Clinical Practice Guideline for circadian rhythm sleep-wake disorders recommends a combination of behavioral, light, and pharmacologic approaches. Treatment aims to shift the body clock earlier (phase advance) and then keep it stable.
1. Strategically Timed Light Exposure
Bright light in the biological morning — meaning shortly after the individual's natural wake time, not at an arbitrary clock time — is the most powerful phase-advancing intervention.
- Light intensity: 2,000–10,000 lux (a commercial 10,000-lux light therapy box at 30–60 cm, or direct outdoor sunlight, which is 10,000–100,000 lux even on overcast days).
- Duration: 30 minutes daily.
- Timing: Begin at the current wake time; as the clock advances, gradually move the light exposure earlier (about 15 minutes every 1–2 days).
- Important: Light received before the core body temperature minimum will delay the clock further, making DSPS worse. Timing matters more than intensity.
2. Strategically Timed Melatonin
Low-dose melatonin acts as a chronobiotic, nudging the clock earlier when taken in the biological evening. This is the opposite of how melatonin is often used (as a sleep aid right before bed).
- Dose: Most evidence supports 0.3 to 0.5 mg — much lower than the 3–10 mg doses typically sold in pharmacies. Higher doses can cause grogginess and are not more effective.
- Timing: Approximately 5–7 hours before the desired bedtime (which is roughly 2 hours before the current DLMO).
- Once the schedule stabilizes, the dose can often be tapered.
Melatonin and timed light are synergistic — their effects add together, and the AASM guideline notes that combining them is more effective than either alone in many patients.
3. Consistent Sleep and Wake Times
A fixed wake time — enforced seven days a week — is the backbone of any circadian treatment. Sleeping in on weekends erases gains made during the week ("social jet lag"). This is usually the single hardest behavioral change, and it is non-negotiable.
4. Minimize Evening Light
Dim all lights 2–3 hours before the target bedtime. Reduce screen exposure or use blue-light-blocking glasses in the last hour. Avoid bright bathroom lights for middle-of-the-night trips.
5. Chronotherapy (Rarely Used Today)
A legacy approach in which the patient progressively delays sleep by 2–3 hours each day around the clock until they land at the desired bedtime. This works on the principle that it is easier to delay the clock than to advance it. It requires complete isolation from social and occupational demands for about a week and is now rarely recommended, both because of its impracticality and because it can destabilize the circadian system and trigger Non-24-Hour Sleep-Wake Disorder in susceptible individuals.
6. Treat Comorbidities
Depression, anxiety, and other sleep disorders frequently travel with DSPS. Effective treatment of DSPS often improves mood — and treating depression often makes DSPS treatment more successful.
What to Expect During Treatment
- Most patients see meaningful shifts within 2–6 weeks when light, melatonin, and a fixed wake time are used together.
- Progress is measured in 15–30 minute shifts per day, not hours overnight.
- Relapse is common if the program is stopped abruptly — ongoing maintenance (consistent wake time, morning light exposure) is typically needed long-term.
- Expect temporary sleep debt in the first two weeks, since the old late sleep time and the new early wake time compress sleep until the body clock catches up.
When to See a Sleep Specialist
Consider a referral to a sleep medicine specialist if:
- You have tried consistent schedules, morning light, and over-the-counter melatonin for several weeks without improvement.
- You have significant daytime impairment (missed work or school, car accidents, depression).
- Symptoms started suddenly or are progressing.
- You suspect comorbid sleep apnea, narcolepsy, or another sleep disorder.
- You are considering prescription medication or chronotherapy.
A sleep specialist can arrange actigraphy, measure DLMO, rule out mimics, and develop an individualized treatment plan.
Key Takeaways
- DSPS is a real biological disorder of the body clock, not poor discipline or laziness.
- The master clock lives in the SCN and is synchronized primarily by light reaching melanopsin-containing retinal ganglion cells.
- The SCN controls melatonin release from the pineal gland — light suppresses melatonin, darkness permits it.
- In DSPS, the entire circadian cycle — sleep, alertness, core temperature, melatonin — is shifted several hours later than desired.
- Diagnosis is clinical and usually confirmed with a sleep log or actigraphy for 1–2 weeks.
- Treatment centers on strategically timed bright light in the morning, low-dose melatonin in the biological evening, a fixed wake time, and minimizing evening light.
- DSPS responds well to treatment, but maintenance is essential — the clock will drift later again if the program is abandoned.